icotec
The Spine Tumor Company™
We Help Patients
The world’s first spinal implants made of nonmetallic and radiolucent BlackArmor® enables a full spectrum of treatment modalities in adjuvant tumor therapy and enhances postoperative imaging diagnostics.
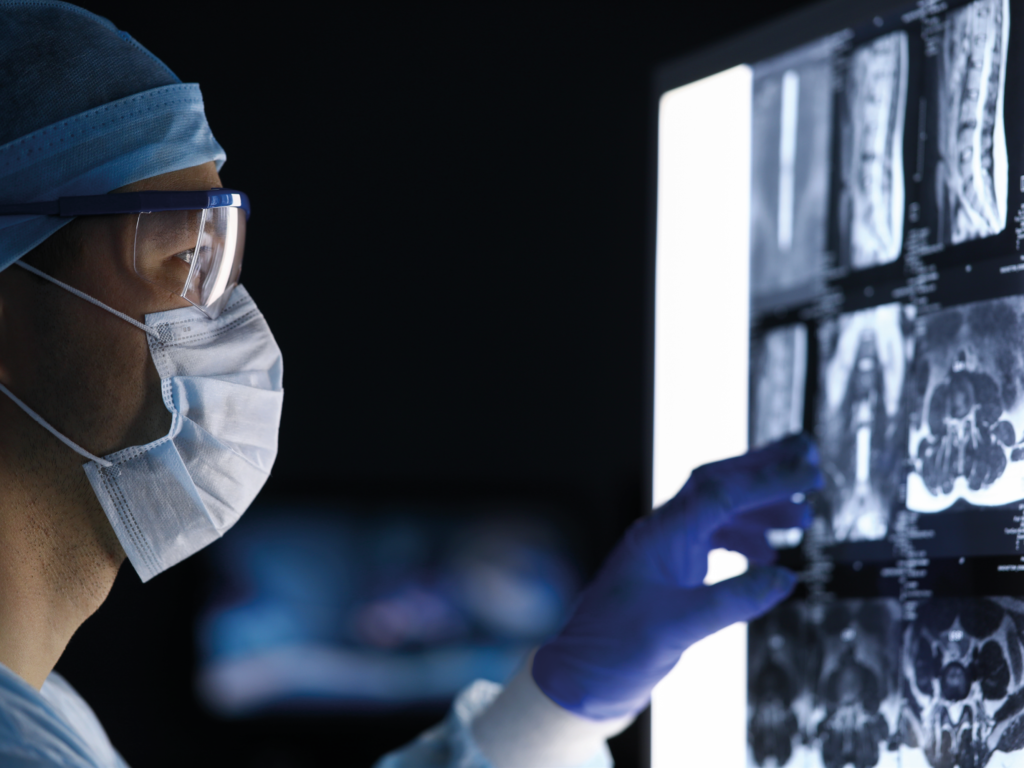
Learn How BlackArmor® Implants Improve the Treatment of Patients with Spinal Neoplasms
Crucial benefits along the entire treatment path: optimized radiation therapy, expanded diagnostic and therapeutic options, and reliable follow-up.

Patients and Caregivers
Being diagnosed with a spinal tumor is challenging and can turn your life upside down. Understanding your condition and the different therapies available to you is the first step on your way Back to Life. BlackArmor® can help you on your way Back to Life.
CuttingEdge Technology in the Fight against Spinal Tumors
BlackArmor® Carbon/PEEK from icotec: the ideal material for optimizing care for patients with spinal neoplasms

Literature
Discover more information about our products, browse our
brochures and flyers, and explore links to scientific publications.





