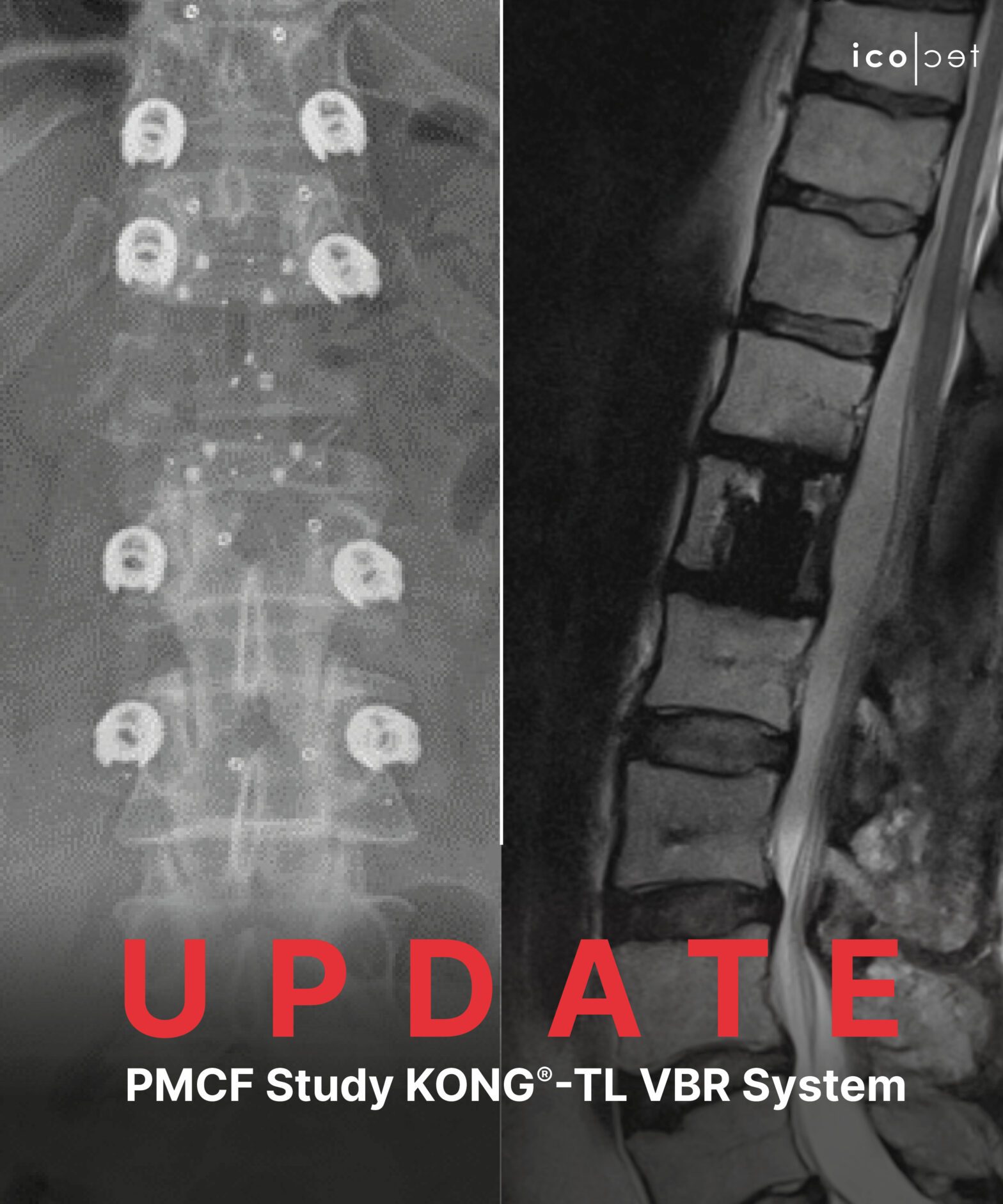
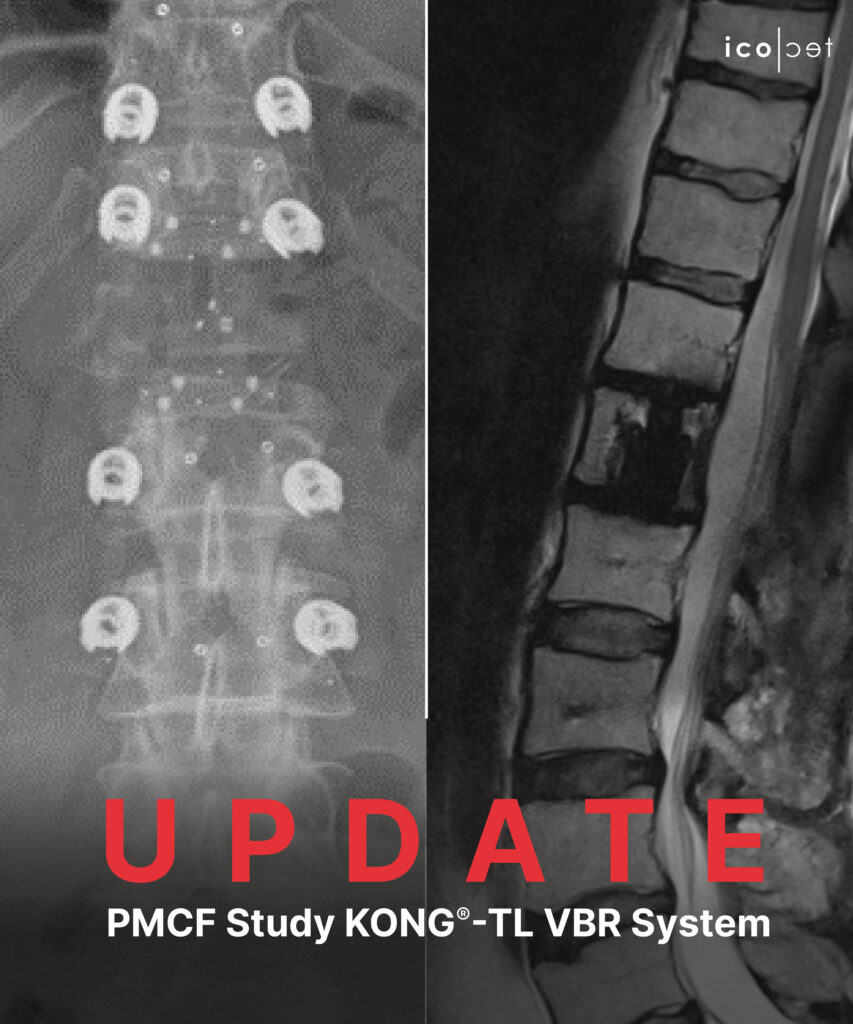
June 1, 2023 – Altstaetten, Switzerland – icotec ag is proud to announce the completion of 50 % patient enrollment for its ongoing multi-center post-market clinical follow-up (PMCF) study with the KONG®-TL VBR System.
This prospective, multi-center, PMCF is focused on investigating the performance and safety of the KONG®-TL VBR System in patients requiring corpectomy procedures followed by vertebral body replacement in the thoracolumbar spine, for tumors or vertebral fractures.
Multiple European hospitals have confirmed patient enrollment in accordance with the study plan, with the anticipation of completing the enrollment process by mid-2024.
We extend our gratitude to the dedicated staff at icotec and the study centers for their meticulous data collection and study administration over the planned 24-month follow-up period.
Our KONG®-TL VBR expandable corpectomy cage is made from BlackArmor® Carbon/PEEK which provides artifact-reduced postoperative imaging combined with reliable osseointegration facilitated by our unique Ti-iT® titanium coating. Tumor patients further benefit from reliable radiation therapy planning and application thanks to our radiolucent material. Stay tuned as we eagerly anticipate sharing the initial results.
About icotec ag
Founded in 1999, icotec ag is a family-owned SME based in Altstaetten, Switzerland. icotec develops, manufactures, and distributes spinal implants made from radiolucent and artifact-free BlackArmor®. icotec’s proprietary BlackArmor® material is made up of continuous carbon fibers combined with PEEK and is processed with icotec’s unique manufacturing technology. With 20 years of clinical success and more than 50,000 implanted products, icotec ag is a pioneer in cutting-edge spinal tumor therapy. For further information, visit our website www.icotec-medical.com or contact us at media@icotec-medical.com.